-
所属单位:
山东大学
-
发表刊物:
Biotechnol Biofuels
-
关键字:
Aspergillus niger, GH11 xylanases, Transcription analysis, Degradation pattern, Synergistic hydrolysis
-
摘要:
Background: Xylan is the most abundant hemicellulose polysaccharide in nature, which can be converted into high value-added products. However, its recalcitrance to breakdown requires the synergistic action of multiple enzymes. Aspergillus niger, possessing numerous xylan degrading isozyme-encoding genes, are highly efective xylan degrad�ers in xylan-rich habitats. Therefore, it is necessary to explore gene transcription, the mode of action and cooperation mechanism of diferent xylanase isozymes to further understand the efcient xylan-degradation by A. niger.
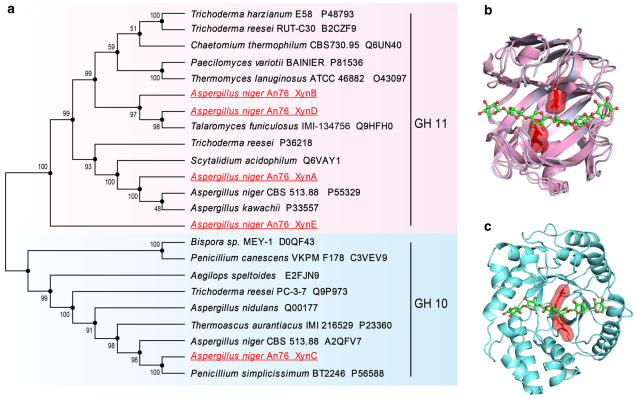
Results: Aspergillus niger An76 encoded a comprehensive set of xylan-degrading enzymes, including fve endo-xyla�nases (one GH10 and four GH11). Quantitative transcriptional analysis showed that three xylanase genes (xynA, xynB and xynC) were up-regulated by xylan substrates, and the order and amount of enzyme secretion difered. Specifcally, GH11 xylanases XynA and XynB were initially secreted successively, followed by GH10 xylanase XynC. Biochemical analyses displayed that three GH11 xylanases (XynA, XynB and XynD) showed diferences in catalytic performance and product profles, possibly because of intricate hydrogen bonding between substrates and functional residues in the active site architectures impacted their binding capacity. Among these, XynB had the best performance in the degradation of xylan and XynE had no catalytic activity. Furthermore, XynA and XynB showed synergistic efects during xylan degradation.
Conclusions: The sequential secretion and diferent action modes of GH11 xylanases were essential for the efcient xylan degradation by A. niger An76. The elucidation of the degradation mechanisms of these xylanase isozymes further improved our understanding of GH-encoding genes amplifcation in flamentous fungi and may guide the design of the optimal enzyme cocktails in industrial applications.
背景:木聚糖是自然界中最丰富的半纤维素多糖,可以转化为高附加值的产品。然而,其对分解的抵抗要求多种酶的协同作用。具有许多木聚糖降解同工酶编码基因的黑曲霉是富含木聚糖的栖息地中高效的木聚糖降解剂。因此,有必要探索不同木聚糖酶同工酶的基因转录,作用方式和协同机制,以进一步了解黑曲霉有效的木聚糖降解作用。
结果:黑曲霉An76编码了一套完整的木聚糖降解酶,包括五个内木聚糖酶(一个GH10和四个GH11)。定量转录分析表明,木聚糖底物上调了三个木聚糖酶基因(xynA,xynB和xynC),并且酶的分泌顺序和数量有所不同。具体地,首先连续分泌GH11木聚糖酶XynA和XynB,随后是GH10木聚糖酶XynC。生化分析表明,三种GH11木聚糖酶(XynA,XynB和XynD)显示出催化性能和产物分布的差异,这可能是由于底物与活性位点结构中功能残基之间复杂的氢键影响了它们的结合能力。其中,XynB在木聚糖降解方面具有最佳性能,而XynE没有催化活性。此外,XynA和XynB在木聚糖降解期间显示出协同效应。
结论:GH11木聚糖酶的顺序分泌和不同的作用方式对于黑曲霉An76有效地降解木聚糖至关重要。这些木聚糖酶同工酶降解机制的阐明进一步增进了我们对火焰真菌中GH编码基因扩增的理解,并可能指导工业应用中最佳酶混合物的设计。
-
论文类型:
期刊论文
-
学科门类:
理学
-
一级学科:
生物学
-
文献类型:
J
-
卷号:
14
-
期号:
118
-
影响因子:
5.473
-
DOI码:
10.1186/s13068-021-01967-1
-
是否译文:
否
-
发表时间:
2021-03
-
收录刊物:
SCI
摘要:
Background: Xylan is the most abundant hemicellulose polysaccharide in nature, which can be converted into high value-added products. However, its recalcitrance to breakdown requires the synergistic action of multiple enzymes. Aspergillus niger, possessing numerous xylan degrading isozyme-encoding genes, are highly efective xylan degrad�ers in xylan-rich habitats. Therefore, it is necessary to explore gene transcription, the mode of action and cooperation mechanism of diferent xylanase isozymes to further understand the efcient xylan-degradation by A. niger.
Results: Aspergillus niger An76 encoded a comprehensive set of xylan-degrading enzymes, including fve endo-xyla�nases (one GH10 and four GH11). Quantitative transcriptional analysis showed that three xylanase genes (xynA, xynB and xynC) were up-regulated by xylan substrates, and the order and amount of enzyme secretion difered. Specifcally, GH11 xylanases XynA and XynB were initially secreted successively, followed by GH10 xylanase XynC. Biochemical analyses displayed that three GH11 xylanases (XynA, XynB and XynD) showed diferences in catalytic performance and product profles, possibly because of intricate hydrogen bonding between substrates and functional residues in the active site architectures impacted their binding capacity. Among these, XynB had the best performance in the degradation of xylan and XynE had no catalytic activity. Furthermore, XynA and XynB showed synergistic efects during xylan degradation.
Conclusions: The sequential secretion and diferent action modes of GH11 xylanases were essential for the efcient xylan degradation by A. niger An76. The elucidation of the degradation mechanisms of these xylanase isozymes further improved our understanding of GH-encoding genes amplifcation in flamentous fungi and may guide the design of the optimal enzyme cocktails in industrial applications.
背景:木聚糖是自然界中最丰富的半纤维素多糖,可以转化为高附加值的产品。然而,其对分解的抵抗要求多种酶的协同作用。具有许多木聚糖降解同工酶编码基因的黑曲霉是富含木聚糖的栖息地中高效的木聚糖降解剂。因此,有必要探索不同木聚糖酶同工酶的基因转录,作用方式和协同机制,以进一步了解黑曲霉有效的木聚糖降解作用。
结果:黑曲霉An76编码了一套完整的木聚糖降解酶,包括五个内木聚糖酶(一个GH10和四个GH11)。定量转录分析表明,木聚糖底物上调了三个木聚糖酶基因(xynA,xynB和xynC),并且酶的分泌顺序和数量有所不同。具体地,首先连续分泌GH11木聚糖酶XynA和XynB,随后是GH10木聚糖酶XynC。生化分析表明,三种GH11木聚糖酶(XynA,XynB和XynD)显示出催化性能和产物分布的差异,这可能是由于底物与活性位点结构中功能残基之间复杂的氢键影响了它们的结合能力。其中,XynB在木聚糖降解方面具有最佳性能,而XynE没有催化活性。此外,XynA和XynB在木聚糖降解期间显示出协同效应。
结论:GH11木聚糖酶的顺序分泌和不同的作用方式对于黑曲霉An76有效地降解木聚糖至关重要。这些木聚糖酶同工酶降解机制的阐明进一步增进了我们对火焰真菌中GH编码基因扩增的理解,并可能指导工业应用中最佳酶混合物的设计。